What is a heat pump?
Heat, by nature, always moves from an area of higher temperature to an area of lower temperature. This “natural” heat transfer takes place constantly all around us. Examples include:
• Heat leaving our skin and clothing surfaces, and transferring to cooler air surrounding us.
• Heat transferring from the inside of buildings to outside air whenever the inside temperature is warmer than the outdoor temperature.
• A glass of cold iced tea placed on a countertop continually absorbing heat from warmer air surrounding it, as well as from the countertop, both of which are at higher temperature than the tea.
No machines or special techniques are needed to move heat from materials at higher temperature to materials at lower temperature.
Heat pumps were developed to reverse the “natural” direction of heat transfer. Their function is to move (e.g., “pump”) heat from materials at lower temperature to materials at higher temperature.
The low-temperature heat is gathered from some material called the “source,” and then concentrated and released into another material called the “sink.”
In some respects, a heat pump is similar to a refrigerator. The latter absorbs low-temperature heat from the food placed inside it. It then raises the temperature of the absorbed heat and releases it in the surrounding air. Most heat pumps and refrigerators use a chemical called a refrigerant that circulates within a closed circuit of components and changes phase between liquid and vapor to “pump” heat from low-temperature materials into higher-temperature materials. The refrigerant is pushed through the closed loop of components by an electrically operated compressor. The details of this refrigeration cycle are discussed in section 2.
Although there are similarities between heat pumps and refrigerators, there are also very distinct differences. Most heat pumps are designed to operate at higher rates of heat transfer compared to a common refrigerator. Most heat pumps can also reverse which material supplies the low-temperature heat and which material receives the higher-temperature heat. This makes it possible for heat pumps to heat and cool buildings. There are also many different configurations of heat pumps available depending on the material from which low-temperature heat is being absorbed, and the material into which higher-temperature heat is being released.
When used to heat buildings, heat pumps can gather low-temperature heat from sources such as outdoor air, ground water, lakes or ponds, or tubing buried in the earth. All of these sources provide “free” low-temperature heat.
Heat pumps that extract low-temperature heat from outside air are common in North America. They are appropriately called “air-source” heat pumps. The vast majority of air-source heat pumps currently in service are configured to deliver higher-temperature heat through a forced-air distribution system within the building. This leads to the more specific classification of “air-to-air” heat pump.
Heat pumps that extract low-temperature heat from geothermal sources such as lakes, ponds, wells or tubing buried in the earth use water or an antifreeze solution to convey heat from those sources to the heat pump. They are thus classified as water-source heat pumps. Water-source heat pumps that deliver heat through a forced-air system are more specifically called “water-to-air” heat pumps. Those that deliver heat using a hydronic distribution system are known as “water-to-water” heat pumps.
This issue of idronics deals with a specific heat pump configuration that absorbs low-temperature heat from outside air and delivers that heat, at higher temperatures, to a stream of water within a building. This type of heat pump is more specifically called an “air-to-water” heat pump.
History of Heat Pumps
Heat pumps are based on the principles of refrigeration, which were first demonstrated by Scottish physician William Cullen in 1755. Cullen developed an apparatus to create a vacuum over a container of ether immersed in water. The vacuum caused the ether to boil, and in doing so, absorb heat from the water to create a small amount of ice.
The thermodynamic principles underlying heat pumps are partially credited to Lord Kelvin, who contributed to the formulation of the first and second laws of thermodynamics and proposed the concept of an absolute temperature scale. The French engineer Sadi Carnot also contributed to the thermodynamic underlying “heat engines,” which are devices that extract energy from some higher-temperature material and convert that energy into a combination of mechanical work and lower-temperature heat. From a thermodynamic perspective, a heat pump can be thought of as a heat engine operating in reverse. It combines heat from a low-temperature source material with mechanical work to produce heat at a higher temperature. Carnot, building on the work of Kelvin, also developed a formula that sets the theoretical performance limits for any heat pump.
Crude heat pumps were developed in the early 1900s but remained little more than science experiments at a time when fossil fuels, especially coal and petroleum, were the dominant energy source for heating buildings.
The first heat pumps to be mass produced were based on machines used for central air conditioning. The Carrier Corporation is widely recognized as one of the first companies to commercialize residential central cooling using vapor-compression refrigeration systems. During the 1950s, Carrier Corporation provided over 700 early-generation central air-conditioning systems for one of the first large-scale housing developments in Levittown, Pennsylvania.
Although often taken for granted today, the advent of central air conditioning at that time allowed scarcely populated areas in the southwestern U.S. to develop into major population centers. Some historians have even cited air conditioning as one of the most impactful technical accomplishments of the 20th century.
Early-generation air-conditioning systems were only able to cool buildings, absorbing heat from interior spaces and dissipating it to outside air. The next technological hurdle was finding ways to reverse the direction of heat flow, and thus convert low-temperature heat in outside air into higher-temperature heat to maintain comfort in buildings. This was the advent of air-to-air heat pumps.
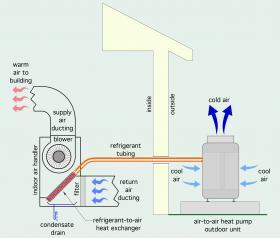
A basic "split system"
The basic configuration of a “split system” air-to-air heat pump, operating in heating mode, is shown in Figure 1-1.
The outside unit, often called the “condenser” because of its origin in air-conditioning systems, connects to an interior air handler using two copper refrigerant tubes. The compressor is located in the outdoor unit. The indoor unit contains a refrigerant-to-air heat exchanger and blower.
Early air-to-water heat pump experiences.
As is often the case with new technologies, early experiences with air-to-air heat pumps were mixed. First generation products experienced higher than acceptable compressor failure rates. In 1964, this reliability issue led he U.S. Department of Defense to issue a ban on the use of heat pumps in military facilities due to the severity of maintenance problems. Fossil fuel continued to be the dominant energy source for heating buildings.
The OPEC oil embargo, which began in 1973, reinvigorated efforts to develop reliable electrically powered heat pumps that could lessen dependence on petroleum-based heating fuels. Manufacturers of air-conditioning systems worked on methods of reversing the direction of heat flow, and thus allow low-temperature heat in outside air to be raised to temperatures sufficient for heating buildings.
The OPEC oil embargo, which began in 1973, reinvigorated efforts to develop reliable electrically powered heat pumps that could lessen dependence on petroleum-based heating fuels. Manufacturers of air-conditioning systems worked on methods of reversing the direction of heat flow, and thus allow low-temperature heat in outside air to be raised to temperatures sufficient for heating buildings.
One of the earliest attempts at creating a vapor-compression machine that could heat as well as cool simply reversed the direction of the entire air conditioner within an opening in an exterior wall. Another used multiple dampers to change airflow directions.
Manufacturers eventually discovered that the refrigerant flows used in vapor-compression air conditioners could be reversed using a combination of four hand-operated valves. In time, this approach was replaced by two valves operated by electrical solenoids. Further development led to a single 4-port, electrically operated "reversing valve." This type of valve, which is discussed in more detail later in this issue, is now used in a wide variety of heat pumps that provide heating and cooling.
Reversing valves made it practical to heat and cool homes using air-to-air heat pumps. Sales of residential air-to-air heat pumps grew rapidly during the 1970s. The primary markets were southern states with relatively mild winter temperatures and a definite need for summer cooling. Air-to-air heat pumps became heavily promoted by southern electric utilities, as well as by manufacturers such as Carrier, Westinghouse and General Electric.
During the 1970s, the reliability of air-to-air heat pumps continually improved through revised compressor design, better lubrication details and techniques to reduce liquid “slugging” of compressors. By 1975, the U.S. Department of Defense lifted their previous ban on heat pumps in military facilities. A surge of interest in heat pumps during 1976 lead to an annual sales growth rate of 96%. Manufacturers were having difficulty keeping up with demand. By 1978, it was estimated that air-to-air heat pumps were installed in over 1.4 million U.S. homes.
Performance Limitations
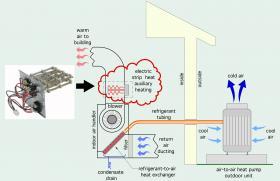
Early generation air-to-air heat pumps could not operate well at the low outdoor temperatures experienced in the Northern U.S. and Canada. Many were limited to minimum outdoor temperatures in the range of 15-20ºF. If the outdoor temperature dropped below this limit, the heat pump would operate at grossly insufficient output or simply turn off. The heating load would then be assumed by electric resistance “strip heaters,” which are heating elements mounted in the supply air plenum on the heat pump’s interior unit, as shown in Figure 1-4.
Strip heat was usually activated by the second stage of a 2-stage thermostat as room air temperature dropped slightly below the desired setting. Although reliable as a supplemental heat source, strip heat, like all electric-resistance heating, is expensive to operate. Some early-generation air-to-air heat pumps were also installed along with gas-fired furnaces that would assume the full heating load if the heat pump could not operate due to low outdoor temperature or some other condition.
The inability to operate at the low outdoor air temperatures experienced in many northern states, and much of Canada, created a stigma that air-source heat pumps were only suitable for heating in mild climates. This limitation was one of the largest factors leading to the emergence of geothermal heat pumps during the 1980s. Because water returning from earth loops, wells or large open bodies of water was always above 32ºF, even when outdoor air temperatures were extremely cold, geothermal heat pumps could provide predictable heating performance in northern climates. In milder climates, geothermal heat pumps also provided higher-efficiency cooling performance compared to early-generation air-source heat pumps. The North American market for geothermal heat pumps has grown steady over the last 30 years, largely driven by the potential for high efficiency and thus lower operating cost.
Cold Climate Air-Source Heat Pumps
As the market for geothermal heat pumps increased over the last 20 years, so did efforts to improve the performance of air-source heat pumps. New refrigeration technologies such as enhanced vapor injection (EVI), variable-speed “inverter” compressors, and electronic expansion valves, none of which were available for use in early-generation heat pumps, now allow modern air-source heat pumps to achieve significantly higher thermal performance at cold outdoor temperatures, in some cases as low as -22ºF.
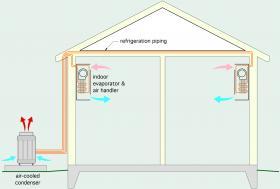
Many “cold climate” air-to-air heat pumps (a.k.a. “low ambient” air-source heat pumps) are currently available as “ductless” split systems. A single outdoor unit connects to one or more indoor air handlers using refrigeration piping. Figure 1-5 shows the concept for a ductless split air-to-air heat pump system with two interior wall-mounted air handlers.
An example of a typical outdoor unit for a modern air-to-air heat pump is shown in Figure 1-6. One of the indoor air handler units connected to this outdoor unit is shown in Figure 1-7.
Although “ductless” split system heat pumps can provide reasonably good thermal performance, they are limited to space heating or cooling. They are also limited by the compromises associated with forced air distribution. These include drafts, dispersal of dust and allergens, potential for clogged air filters, cool air collecting at floor level as warm air rises to ceiling level, possible aggravation of respiratory illnesses and objectionable interior noise.
Modern Air-to-Water Heat Pumps
The same innovations that now make ductless air-to-air heat pumps viable in cold climate applications have been used to create air-to-water heat pumps. When operating in heating mode, these units absorb heat from outdoor air, concentrate that heat to increase its temperature, and transfer it to a stream of water or an antifreeze solution. The heated water can be used for a wide variety of loads such as hydronic space heating, domestic water heating or pool heating. Air-to-water heat pumps can also be reversed to extract heat from an interior stream of water and dissipate it to outside air. As such they can be used to supply several types of chilled-water cooling distribution systems. Modern air-to-water heat pumps provide an ideal combination of low ambient thermal performance along with the unsurpassed comfort and energy efficiency afforded by modern hydronics technology.
One example of a modern air-to-water heat pump is shown in Figure 1-8.
The remainder of this issue will discuss the details for applying modern air-to-water heat pumps in a variety of heating and cooling applications.